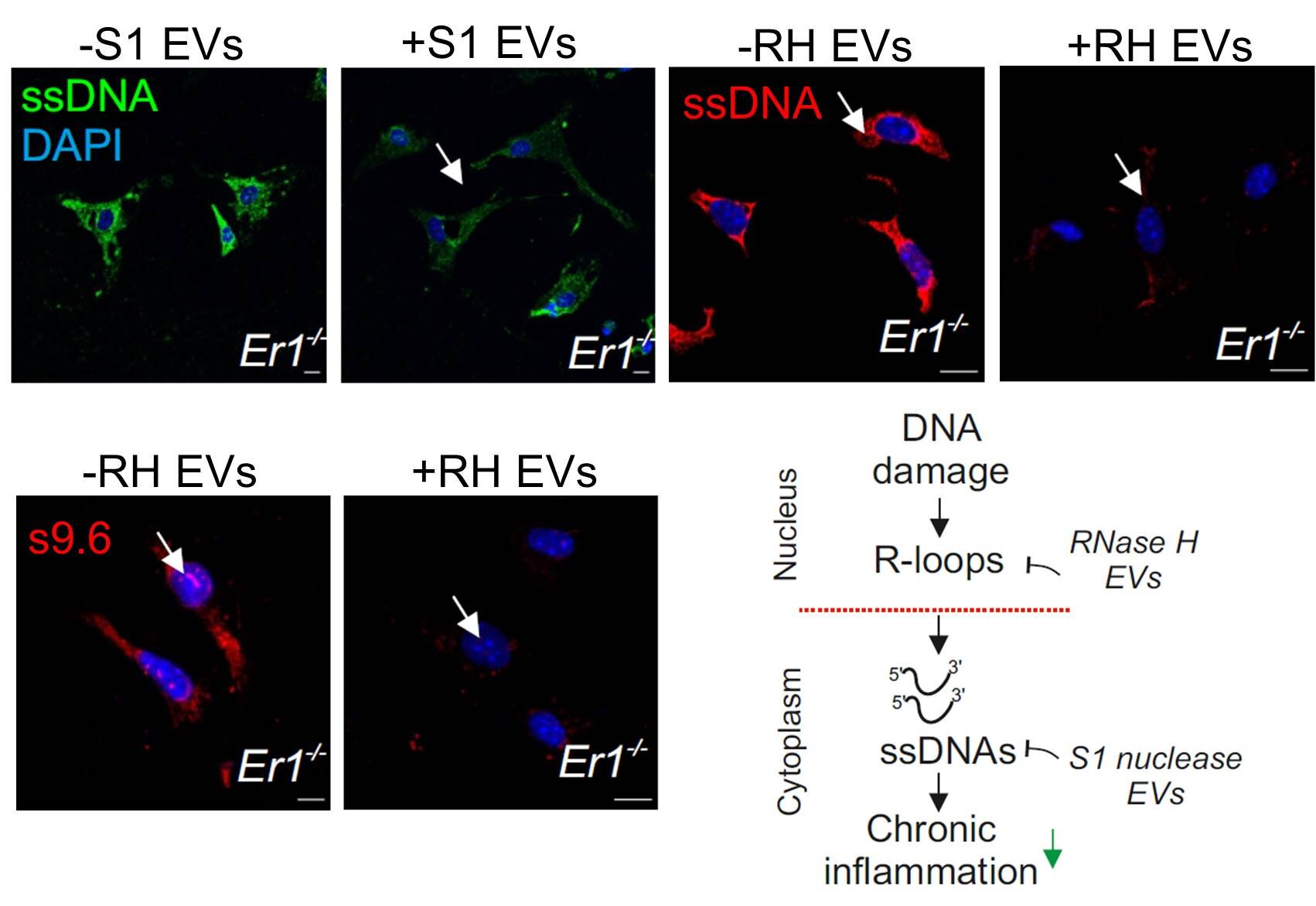
A strategy to deliver ribonuclease H nucleases in inflammatory Ercc1/ pancreatic cells. The treatment of Ercc1/ animals with the EV-delivered nuclease cargo eliminates the effects of damage to the DNA. The Foundation for Research and Technology is based in Hellas.
Research carried out at the Institute of Molecular Biology and Biotechnology (IMBB) of FORTH shows that chronic inflammation and tissue pathology can be caused by persistent DNA damage.
Cancer and aging are associated with inborn defects in DNA repair mechanisms, but also with complex metabolic and endocrine disorders. The researchers at the Institute for Medical and Biological Bladders (IMBB) provide a novel mechanism by which damage to the DNA in animals leads to cellular senescence, fibrosis, loss of tissue architecture and chronic pancreatitis in mice. The team proposed a new therapeutic strategy to combat inflammation and tissue damage associated with aging. The findings show how novel intervention strategies can be used against chronic inflammatory disorders.
The researchers found that the early onset of chronic pancreatitis in the ERCC1-defective animals was caused by the gradual accumulation of irreparable DNA lesions. The release and build-up of single-stranded DNA fragments in the cytoplasm of cells was caused by damage to the DNA hybrid called "R-loops". The immune response in the pancreas of naturally aged mice was stimulated by the cytoplasmic DNA fragments. In order to reduce the pro inflammatory load, the researchers developed an EV-based strategy to deliver RNase H or S1 nuclease. They found that treatment with the EV-delivered nuclease cargo rapidly removes R-loops and the ssDNA moieties in the cytoplasm of pancreatic cells, which reduces the pro inflammatory response seen in the DNA repair deficient mice.
The findings support the idea that the development of EV-based therapeutic regimes against ssDNAs is a promising strategy against inflammation and aging.
The release of ssDNAs leads to chronic inflammation upon DNA damage, thanks to Ourania Chatzidoukaki et al. There is a DOI for this.
Science Advances has journal information.
There is a transcript of the exosome-based metabolism triggered by tissue-infiltrating macrophages.
The document is copyrighted. Any fair dealing for the purpose of private study or research cannot be reproduced without written permission. The content is not intended to be used for anything other than information purposes.
