Figure 1. The cage was engineered with different types of replacements that allowed more IrCp* to be taken up. Takafumi Ueno is from the Tokyo Institute of Technology.
The researchers from Tokyo Tech found that a novel hybrid ferritin nanocage with histidine had 1.5 times higher metal ion absorption and improved catalytic efficiency for alcohol production. Their findings suggest that hybrid bio-nanocages could be used to produce industrial products.
The term "nano-cages" refers to structures that are much smaller than vessels and can be spontaneously self-assembled. These structures can hold a wide range of Molecules that behave as guests. The "ferritin nanocage," which is formed by the self-assembly of 24 subunits into the protein ferritin, is one of the most popular examples. The metal ion helps to convert any substrate into a product. The ferritin cage's potential applications in industry have yet to be fully explored.
Efforts to increase metal ion absorption in ferritin have resulted in cages with low stability. Effective design is the key to making the guest sit in the cage. A team of scientists led by Prof. Takafumi Ueno, from Tokyo Institute of Technology, Japan (Tokyo Tech), introduced site-specific mutations at the core of the ferritin nanocage. Their findings are published in a journal. In the pharmaceutical, food, and chemical industries, iridium is used as a catalyst.
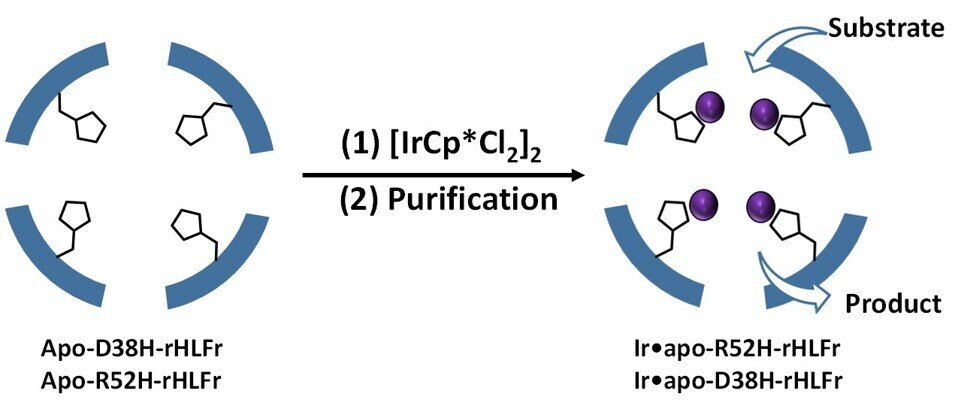
"Based on previous literature, we knew that the presence of coordination amino acids in the cage improve iridium activity, and that substituting these amino acids with appropriate residues could alleviate the problem," says Prof. Ueno. Coordination residues would do the job since iridium complex behaves as a catalyst. The authors used the histidine to replace two of the ferritin cages and created two new ones, R52H and D38H. The cage size was not affected by the changes.
Figure 2. The conversion of substrates to alcohols with high specificity can be done with the help of the nanocage. Takafumi Ueno is from the Tokyo Institute of Technology.
They found that R52H was able to add 1.5 times more iridium atoms than the wild type cage. The D38H Mutant behaved like the wild type. Why didn't both of them have the same effect? Prof. Ueno said that the presence of the histidine residue is important to determine the efficiency of the cage.
The researchers were able to achieve alcohol production rates as high as 88% using the new catalytic cages. The conversion rate was enhanced by a structural re-arrangement of the reaction components.
The researchers used simulations to understand how the substrate behaved inside the cage. There were some interactions between the histidine and the substrate in the R52H Mutant, which were not present in the wild type cage.
Prof. Ueno believes that the hybrid bio-nanocages could be used as catalysts in industrial applications. The current structure-based design of the metal ion binding site research could be improved to create novel ferritin mutants with specific guest molecules.
The study of structure and catalysis is covered in the Angewandte Chemie International Edition. There is a 10.1002/anie.202116623.
The news about cats in a cage was retrieved fromphys.org on January 24, 2022.
The document is copyrighted. Any fair dealing for the purpose of private study or research cannot be reproduced without written permission. The information provided is for educational purposes.