Scientists at KAUST use electron microscopes to investigate the function and structure of key protein complexes. Credit: Heno Hwang.
Cryo-EM has allowed researchers to study how the machinery for replicating DNA assembles at damaged sites.
The cellular DNA is continuously exposed to both the damaging agents and the harmless agents. All living organisms have evolved mechanisms to repair and tolerate DNA damage to try to ensure that genetic information is accurately inherited. Translesion synthesis (TLS) is a mechanism that allows DNA replication to go through unrepaired DNA lesions.
The temporary replacement of highly accurate DNA synthesizing enzymes with specialized, low-fidelity TLS polymerases can ensure cell survival at the expense of introducing mutations. Normal cells can become cancer or drug resistant if they are exposed to the synthetic activity of the TLS polymerases.
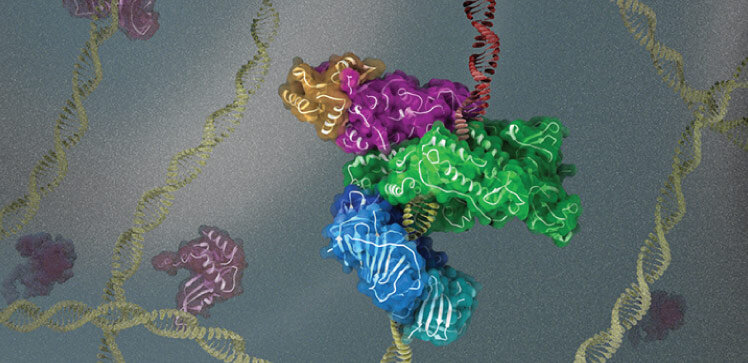
The Y- family TLS polymerase PolK is able to perform DNA synthesis across several damaged bases and is recruited to it by the PCNA. PCNA is regulated by ubiquitination. The addition of a single ubiquitin molecule at the K164 of PCNA facilitates the recruitment and retention of TLS polymerases to damage sites, but the structural basis of the interaction between these polymerases and ubiquitinated PCNA is poorly understood.
The laboratory led by Samir Hamdan, an expert in single-molecule analysis of human DNA replication, has been collaborating with De Biasio's group. They have been using the three-dimensional structure and function of key protein complexes to investigate.
Their latest study describes the reconstructions of full-length human PolK bound to DNA, an incoming nucleotide, and mono-ubiquitylated PCNA at near-atomic resolution. The structure of PolK bound to PCNA is flexible, suggesting that binding to DNA is required to form a complex.
The co-lead author of the study has performed functional studies on how PCNA ubiquitination affects PolK activity.
"Our data gives a framework to explain how PCNA recruits a Y-family TLS polymerase to sites of DNA damage," he says. The PolK-DNA-PCNA complex has a high degree of domain conserved between Y- family polymerases.
Understanding the interactions between the proteins forming these complexes and how they are regulated can help us identify ways to increase or decrease their function for medical applications.
The structure of human Pol bound to DNA and mono-ubiquitylated PCNA is more information. There is a DOI of 10.1038/s41467-021-2
Nature Communications is a journal.
The DNA replication under the microscope was retrieved from the news websitephys.org.
The document is copyrighted. Any fair dealing for the purpose of private study or research cannot be reproduced without written permission. The content is not intended to be used for anything other than information purposes.
